Abstract
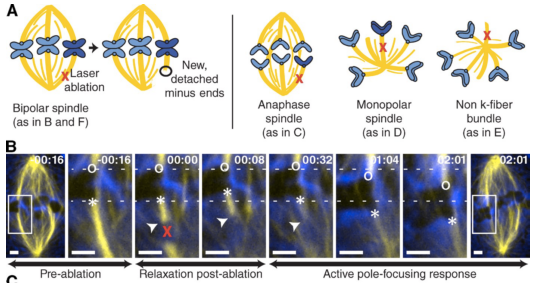
The spindle is a dynamic self-assembling machine that coordinates mitosis. The spindle’s function depends on its ability to organize microtubules into poles and maintain pole structure despite mechanical challenges and component turnover. Although we know that dynein and NuMA mediate pole formation, our understanding of the forces dynamically maintaining poles is limited: we do not know where and how quickly they act or their strength and structural impact. Using laser ablation to cut spindle microtubules, we identify a force that rapidly and robustly pulls severed microtubules and chromosomes poleward, overpowering opposing forces and repairing spindle architecture. Molecular imaging and biophysical analysis suggest that transport is powered by dynein pulling on minus ends of severed microtubules. NuMA and dynein/dynactin are specifically enriched at new minus ends within seconds, reanchoring minus ends to the spindle and delivering them to poles. This force on minus ends represents a newly uncovered chromosome transport mechanism that is independent of plus end forces at kinetochores and is well suited to robustly maintain spindle mechanical integrity.